Scientists turn pure water into metal and it’s captured on video
Scientists managed to convert Water in metal Bright, and although it was for a few seconds, researchers showed for the first time in 2021 that high pressures are not the only conditions that can stimulate mineral In the pure water.
According to the results published in the specialized journal temper naturethe researchers put a A drop of water pure in contact with a alkali metals that share electrons. In this case, it was an alloy of sodium and potassium to which free-moving charged particles could be added, transforming Water in metal.
The resulting conductivity lasts only a a few secondsbut according to the physical Robert SeidelThis result is an important step for understanding this stage of Water Study it directly.
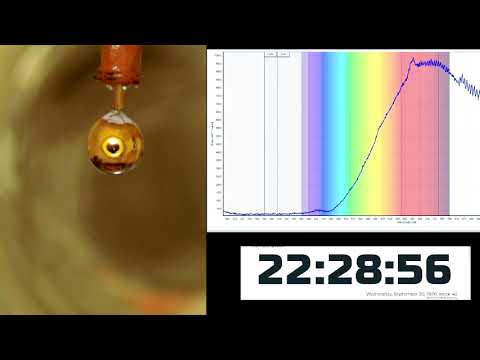
“You can see the phase transition to mineral water with the naked eye! The silver drop of sodium and potassium is covered with a golden sheen, which is impressive.”
Robert Seidel, researcher at Helmholtz-Zentrum Berlin für Materialien und Energie in Germany
Seeing how it can be . says Pavel Jungwirth, a physical chemist at the Czech Academy of Sciences Water He took on a golden glow that was one of the highlights of his career. “It’s a very important advance,” says Peter Edwards, a chemist at the University of Oxford.
Such was the “mineral water” experience!
In a vacuum chamber, the team began to extrude (expand) a small drop of Sodium ingot and potassium, which is liquid at room temperature, and carefully add a thin layer of pure water by deposition steam.
When contacting, electrons and cations metallic flock to Water of the alloy. This not only gives Water A golden glow, but it made it conductive as in pure water metallic under high pressure. This was confirmed using optical reflectance spectroscopy and synchrotron X-ray photoelectron spectroscopy.
Why does this amazing phenomenon occur?
By squeezing the atoms together enough, the orbits of the outer electrons will begin to overlap, allowing them to move. for him Waterthis pressure is about 48 megabars, just under 48 million times the Earth’s atmospheric pressure at sea level.
While pressures beyond this have been created in a laboratory setting, such experiments would not be suitable for studying Mineral water. Therefore, researchers focused on alkali metals. These materials release their outer electrons very easily and can induce exchange properties water electrons Pure ultra pressed without high pressure.
There is a problem turning water into metal!
The alkali metals, like the ones used in the experiment to turn water into metal, is highly reactive with liquid water, sometimes even to the point of exploding. Scientific publications such as the journal Sciences Take a video of him falling in it metal In the Water This sample will explode.
Based on this experience, the scientists added Water to me metal instead of adding metal to me Water. “Our study not only demonstrates the potential for mineral water production on Earth, but also demonstrates the spectral properties associated with its beauty. metallic shine Seidel said.

“Evil coffee nerd. Analyst. Incurable bacon practitioner. Total twitter fan. Typical food aficionado.”

:quality(70):focal(288x128:298x138)/cloudfront-us-east-1.images.arcpublishing.com/metroworldnews/4VWFN4IMGFGQTCCSYSVPIJDM4A.jpg)







