Uganda licenses three experimental treatments for Ebola
Kampala, June 18 (Reuters / EP) –
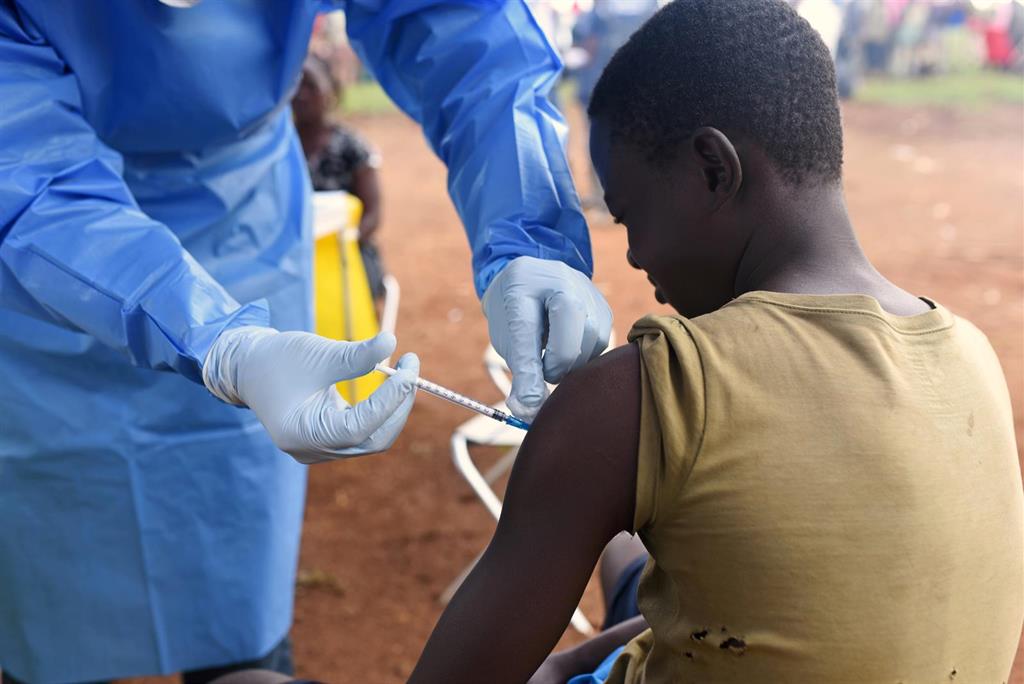
Ugandan health workers have been given the green light to use three experimental treatments for Ebola in the country, a week after the first case of the virus was confirmed on Ugandan territories exported from the neighboring Democratic Republic of the Congo, according to announced reports. the authorities.
Two people from the Democratic Republic of the Congo, a boy and his grandmother, died in Uganda last week. Another three-year-old boy, the brother of the first victim, died after returning to the Democratic Republic of the Congo at the weekend, according to the Congolese Ministry of Health. Another 1,411 people have died in the Democratic Republic of the Congo since August in what is already the second largest worldwide outbreak in history
“We are pleased to inform you that we have received the green light from both the Ugandan National Council for Science and Technology and the National Medicines Authority to bring curative treatment to Ebola patients into the country,” Health Minister Jane Roth wrote on Twitter. .
Authorized treatments are ZMapp, from Mapp Biopharm Pharmaceutical; Regeneron, of Regeneron Pharmaceuticals, and Remdesivir, manufactured by Gilead Sciences, according to World Health Organization (WHO) spokesperson Tarik Jasarevic.
He explained by e-mail that “the protocols for the fourth are being presented. Work is underway to provide logistics services with the support of Doctors Without Borders (MSF) to import a small number of shipments of about ten each.”
The World Health Organization has stated that there are currently no known cases of human-to-human transmission in Uganda, as all patients have traveled from the Democratic Republic of the Congo. Four experimental curative therapies are already in use in this country.
Health workers and people who had contact with infected people in Uganda began receiving an experimental Ebola vaccine from Merck on Saturday.

“Coffee fanatic. Gamer. Award-winning zombie lover. Student. Hardcore internet advocate. Twitter guru. Subtly charming bacon nerd. Thinker.”